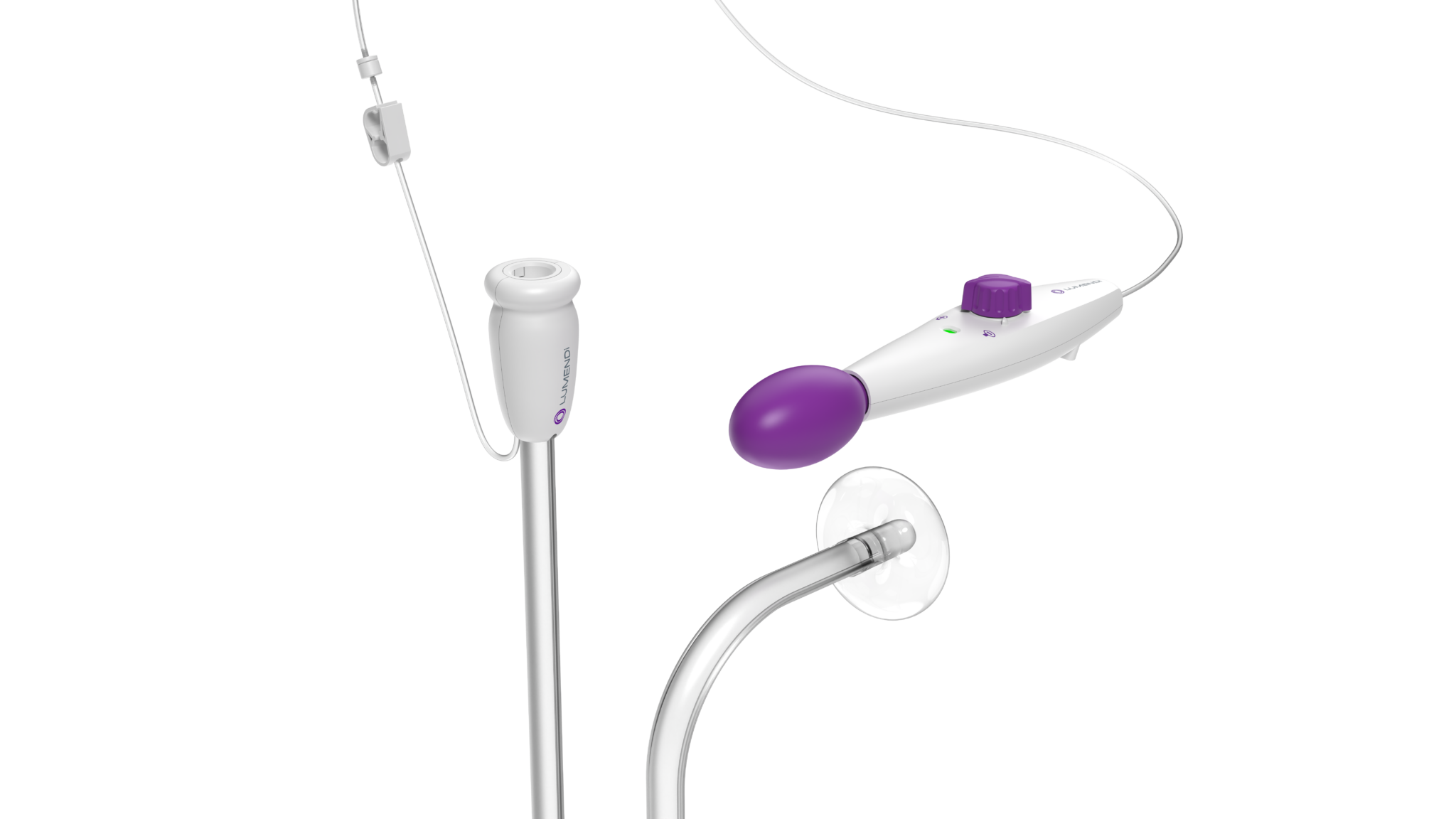
Pioneering endosurgery
Since 2017, Lumendi has worked with gastrointestinal and colorectal practitioners to make complex polypectomies and other GI interventions faster, safer, and less invasive with our innovative DiLumen® Endoluminal Interventional Platform (EIP).
We’re committed to expanding possibilities in endosurgery—helping physicians perform at a higher level and minimize risk and recovery time for patients.
Our products put more possibilities for endosurgery within reach.
DiLumen EZ¹
EZ¹ fits over standard and small-diameter endoscopes used within the lower GI tract.
Learn MoreDiLumen EZ Glide
Enhances EMR, ESD, complex polypectomies, and challenging colonoscopies with superior navigation and improved stability and control.
Learn MoreDiLumen C²
Enabling unprecedented tissue manipulation via robotic-like end effectors to endoluminal surgery in a lower-cost, disposable format.
Learn moreOur latest news & events:
Expand your knowledge of what’s possible in endosurgery with Lumendi.
Leave us your information to stay in touch.