The management team has extensive experience in the healthcare industry and is advised by a group of influential endoscopists.

Peter Johann
Executive Chairman

Pasi Piitunlaninen
CFO

Mike Parrillo
COO

Eric Coolidge
VP Marketing & Sales US

Erwin van Wijk
VP Business Development
Our technology
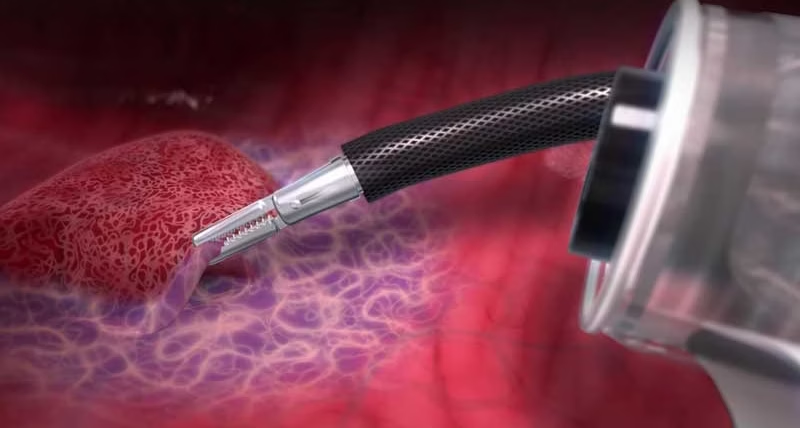
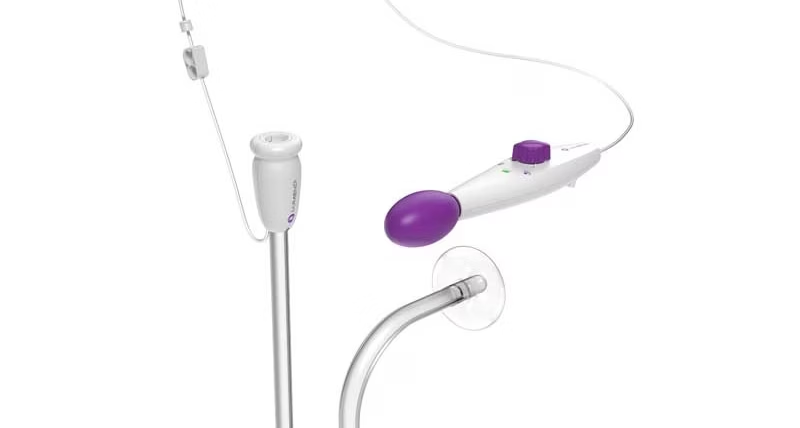
Our innovative DiLumen® Endoluminal Interventional Platform (EIP) technology transformed the endosurgery landscape.
DiLumen works with your existing endoscopic equipment to enable endoluminal therapeutic procedures such as complex polypectomies.
Its two balloons create a stabilized area within the GI tract, improving control and tissue manipulation in the rectum and colon.
Advisory board members comprised of renowned endoscopist that have guided Lumendi’s preoduct development

Mr. Amyn Haji


Dr. Adolfo Para-Blanco


Dr. Sergey Kantsevoy


Dr. David Diehl


Dr. Stavros Stavropoulos


Dr. Emre Gorgun


Dr. Roberta Maselli


Molham Abdulsamad, MD