NCSG annual meeting


“This certification is a key milestone in our global strategy to expand access to minimally invasive endoluminal procedures,” stated Peter Johann, Ph.D., Executive Chairman of Lumendi AG.

Lumendi proudly showcased its DiLumen® portfolio at DDW 2025, emphasizing innovation in advanced colorectal procedures. Our hands-on training sessions highlighted our commitment to physician education and the future of endoluminal care.
At the 2024 Kings Live Event, Lumendi showcased the groundbreaking DiLumen EZ Glide system, demonstrating its superior capabilities in advanced endoscopic procedures through live cases led by Dr. Amyn Haji.
Lumendi sponsored the Florida Third Space Endoscopy Workshop from November 17-19, 2024, at the AdventHealth Nicholson Center, emphasizing its commitment to advancing gastroenterology training. Participants had the opportunity to enhance their skills in Endoscopic Submucosal Dissection (ESD) techniques through hands-on sessions with Lumendi’s innovative interventional platforms.

The prospective, randomized controlled study found that DiLumen reduced total procedure time by 36.5%, leading to an average cost savings of $610.16 per patient.
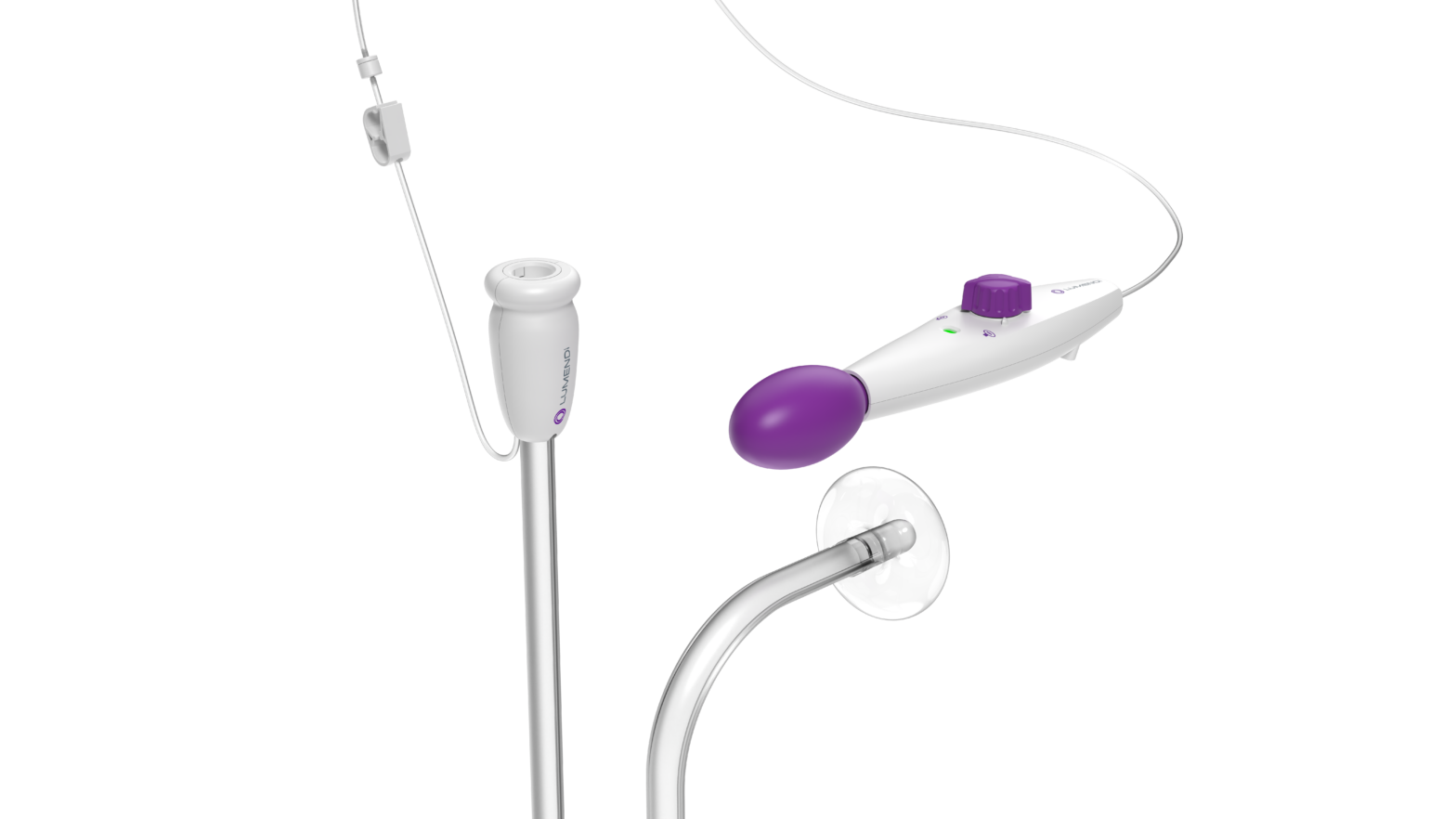
Endoscopic removal of colonic polyps and early cancers eliminates the need for surgical resection and possible temporary or permanent colostomy.

Award for the Balloon Endo-Luminal Lumen Occluding (BELLO) Device Presented to Dr. Maselli at the ESGE Days 2023 Congress Apr 20, 2023, 12:27 ET Lumendi Congratulates Dr. Maselli on the… Continue reading Dr. Roberta Maselli, receives innovation of the year award for the best new device from the ESGE





Since the company’s inception, we have accumulated over 100 DiLumen users worldwide. Half of these physicians have begun using our devices in the last 12 to 18 months. This year’s DDW was our first opportunity in three years to present Lumendi and our significantly enhanced DiLumen platform to users and industry.

“The new DiLumen EZ Glide significantly improved our ability to remove four cecal polyps in a recent case. We could not have ever done this without it.” Dr. Stuart Gordon… Continue reading Dr. Stuart Gordon
“EZ Glide represents a huge improvement in usability of DiLumen technology. The enhanced ease of use has prompted me to use DiLumen more often for complex colonic polypectomies.” Dr. David… Continue reading Geisinger Medical Center, Danville, PA

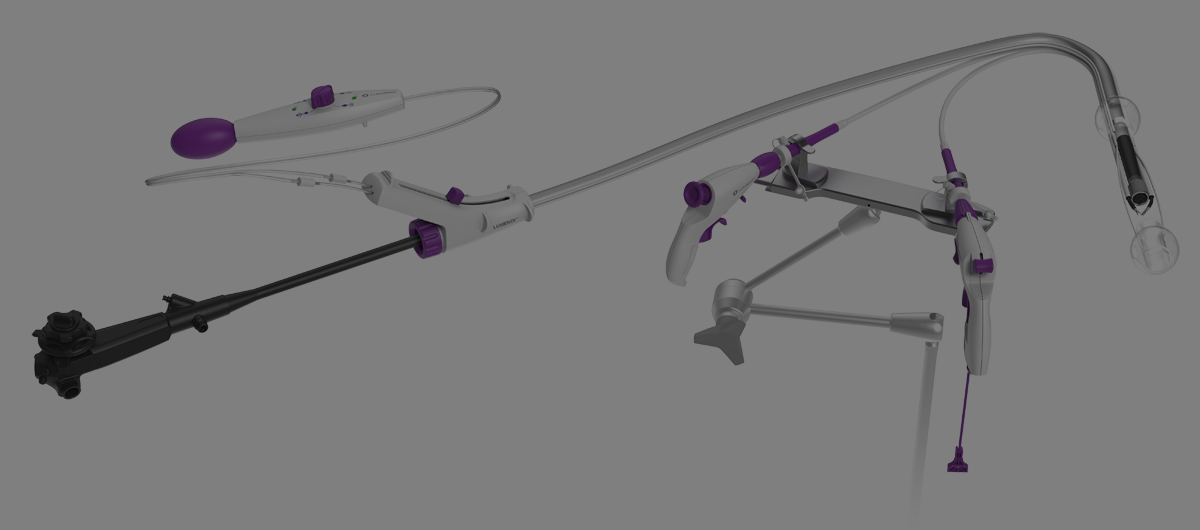
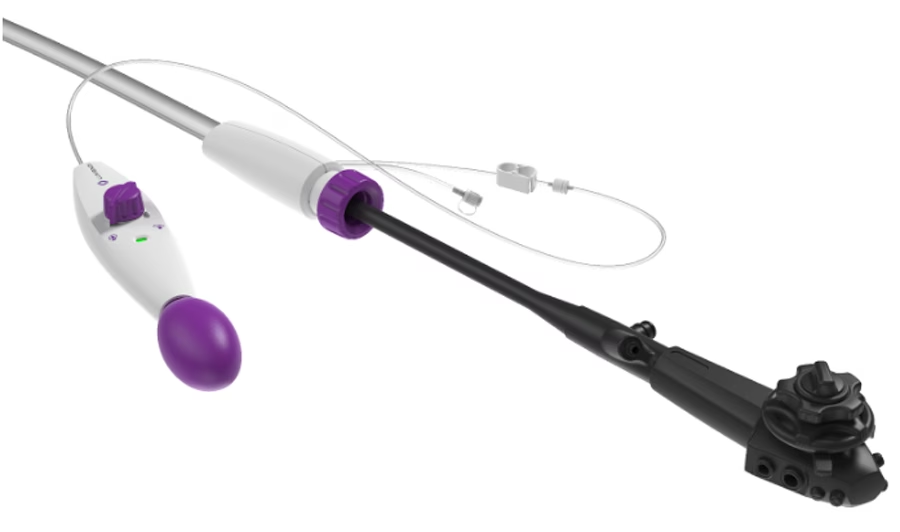
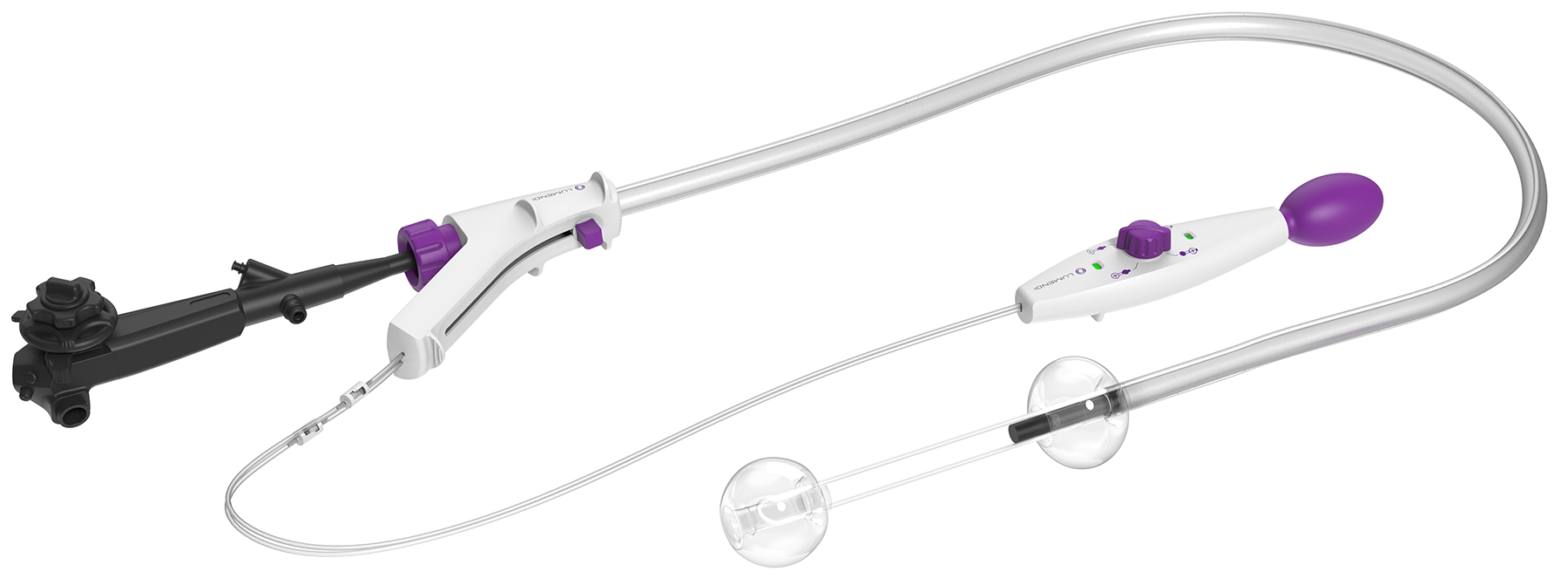
DiLumen EZ-Glide improves visibility and simplifies intraoperative

Company Achieves New Milestone with Over 4,000 Therapeutic Procedures Completed Across the U.S., Europe and Asia, Using DiLumen™ Endoluminal Interventional Platform February 16, 2023 08:00 AM Eastern Standard Time WESTPORT,… Continue reading Lumendi receives 510k clearance for two new devices



Thank you for supporting our Patient Awareness Initiative



Many studies have documented that an endoscopic approach for ESD is a safer and less expensive alternative to removing these polyps, leading to fewer postoperative complications and shorter hospital stays.
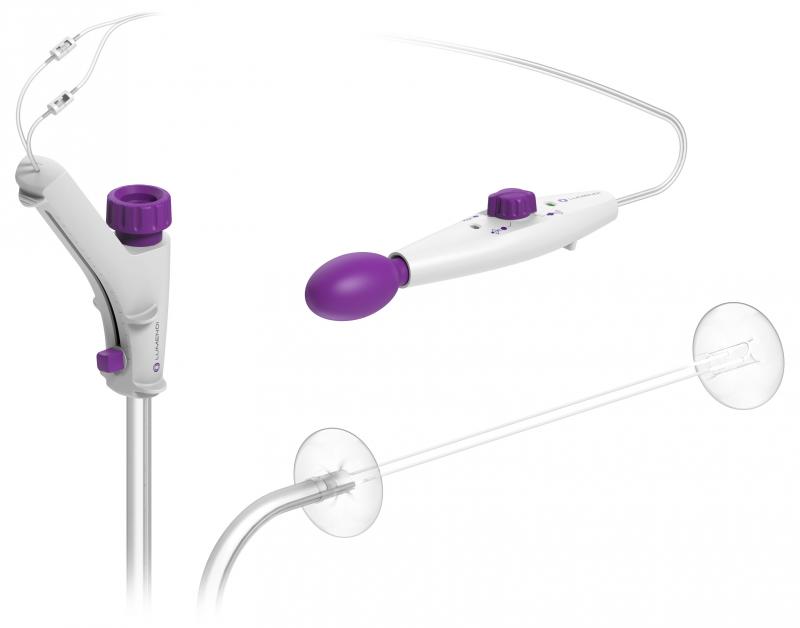



Superiority of DiLumen over traditional (non-overtube) ESD for complex colorectal polypectomy.
Removing colorectal polyps and early cancers by endoscope eliminates the need for surgical resection and the potential for colostomy. This study reports clear improvements in safety, dissection time, dissection speed, R0 resection rates (clean pathological margins), and navigation time over non-DiLumen-assisted (freehand) procedures.
The Cost-Effectiveness of DiLumenTM for Endoscopic Intervention of Complex Colonic Polyps
James White, D.O., VP of Medical Affairs, Lumendi
Karl Florence, Reimbursement Consultant.
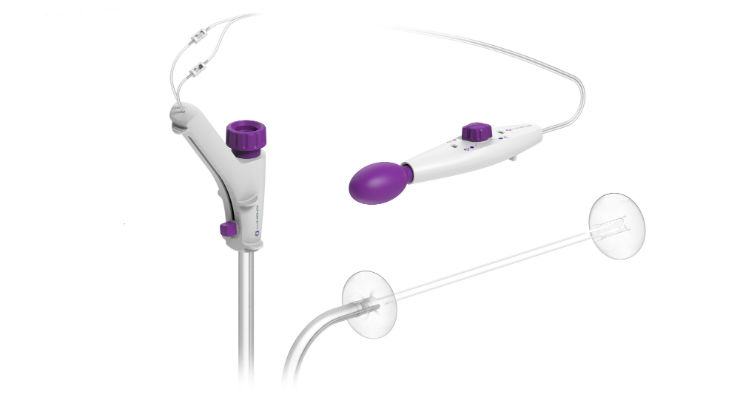
Pioneering Endosurgery reflects Lumendi’s innovative, three-part pathway to:
expand indication areas and procedures in the upper gastrointestinal tract, which includes a recent submission to the FDA;
innovate improvements planned for the DiLumen EIP, including a proprietary lubricated inner surface of DiLumen’s sheath for better endoscope handling performance; and
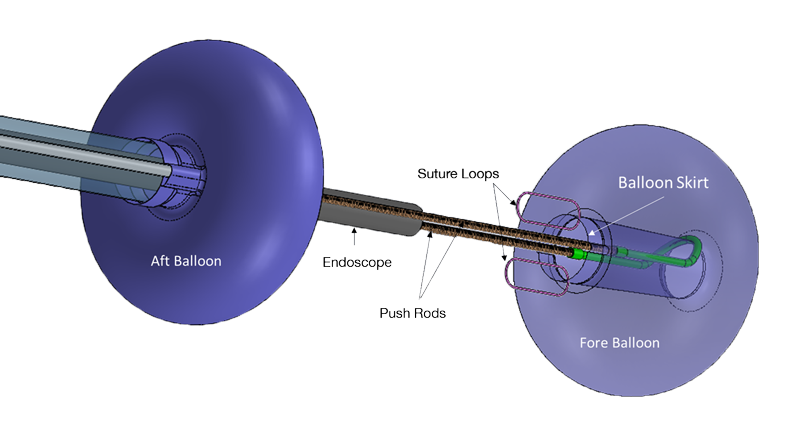
commercially release the DiLumen C2 platform[1] to bring robotic-like end-effectors to endolumenal surgery in a low-cost, disposable format to provide endoscopists with unprecedented control of tissue.

Dr. Kantsevoy states, “Endoscopic endolumenal appendectomy is less invasive than traditional surgical, laparoscopic, transvaginal or transgastric appendectomy.
A study evaluating endoscopic procedure time removing complex colorectal polyps shows superiority of Lumendi’s DiLumen device over a control group.

We have adapted our operation and clinical support models as healthcare facilities prioritize focus on patients affected by COVID-19.

The study evaluates DiLumen’s effect on endoscopic procedure time for pre-cancerous colorectal complex polypectomies.

The first successful case at Cleveland Clinic using DiLumen C2 was completed by Dr. Gorgun when a large complex polyp in the distal sigmoid, 18cm from the anus was removed from a 72-year-old woman with a narrow sigmoid.



With recent regulatory approvals in Europe, Lumendi has established distribution in the UK, France and Italy and has conducted successful procedures with clinicians in each country.